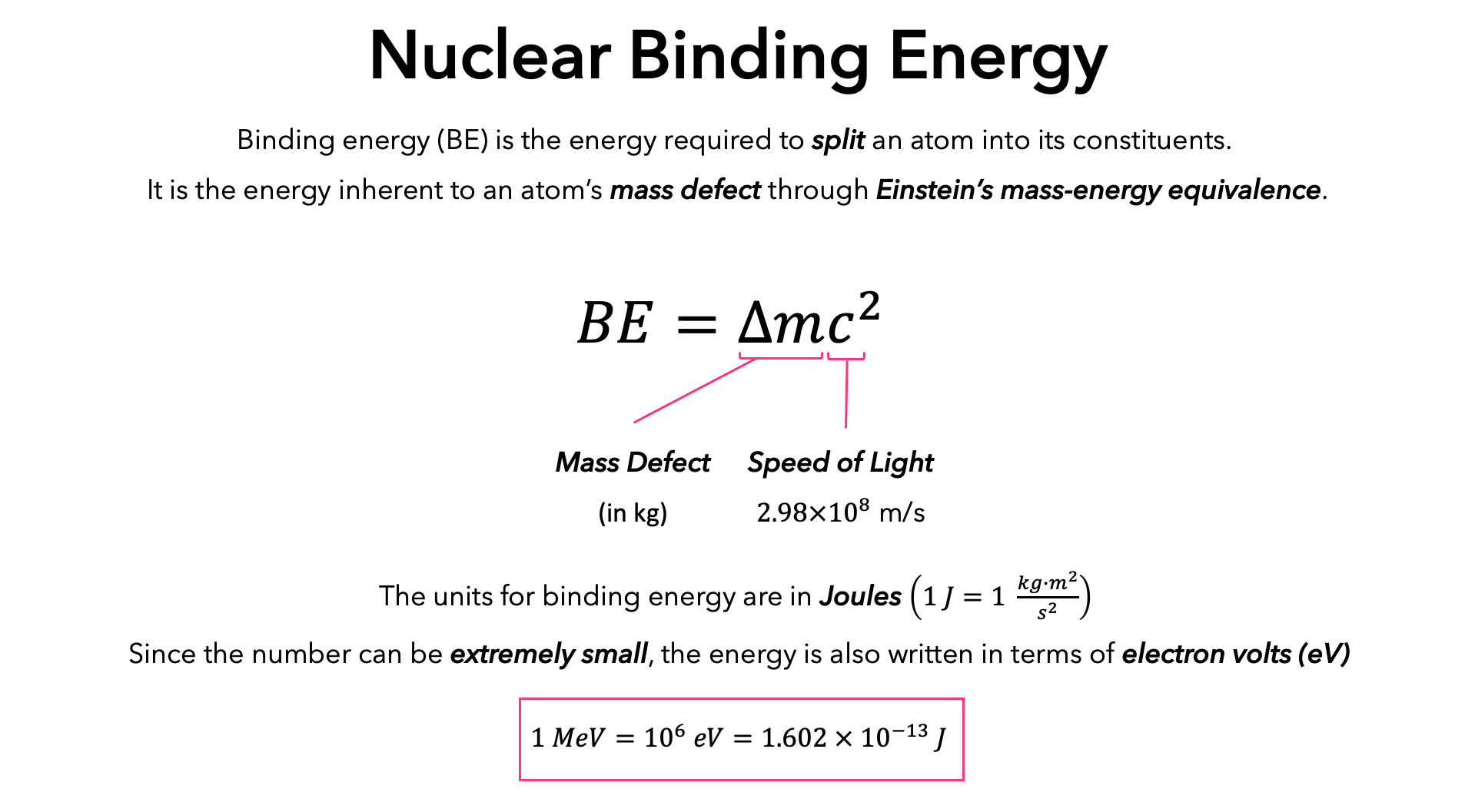
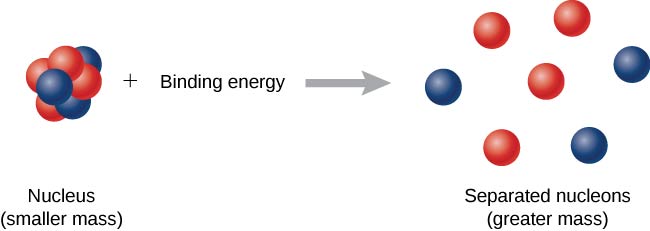
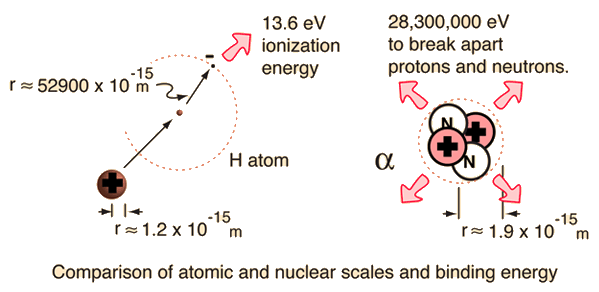
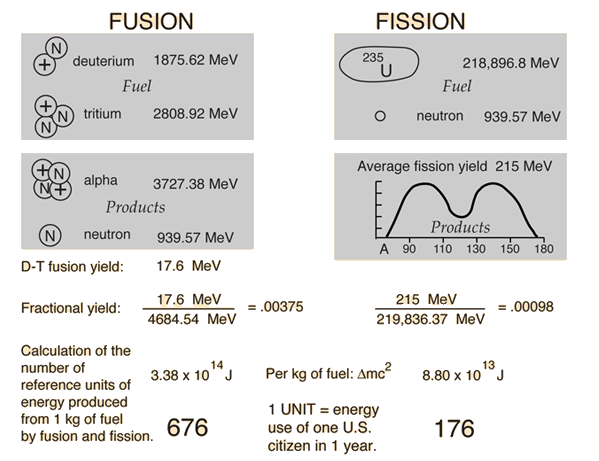
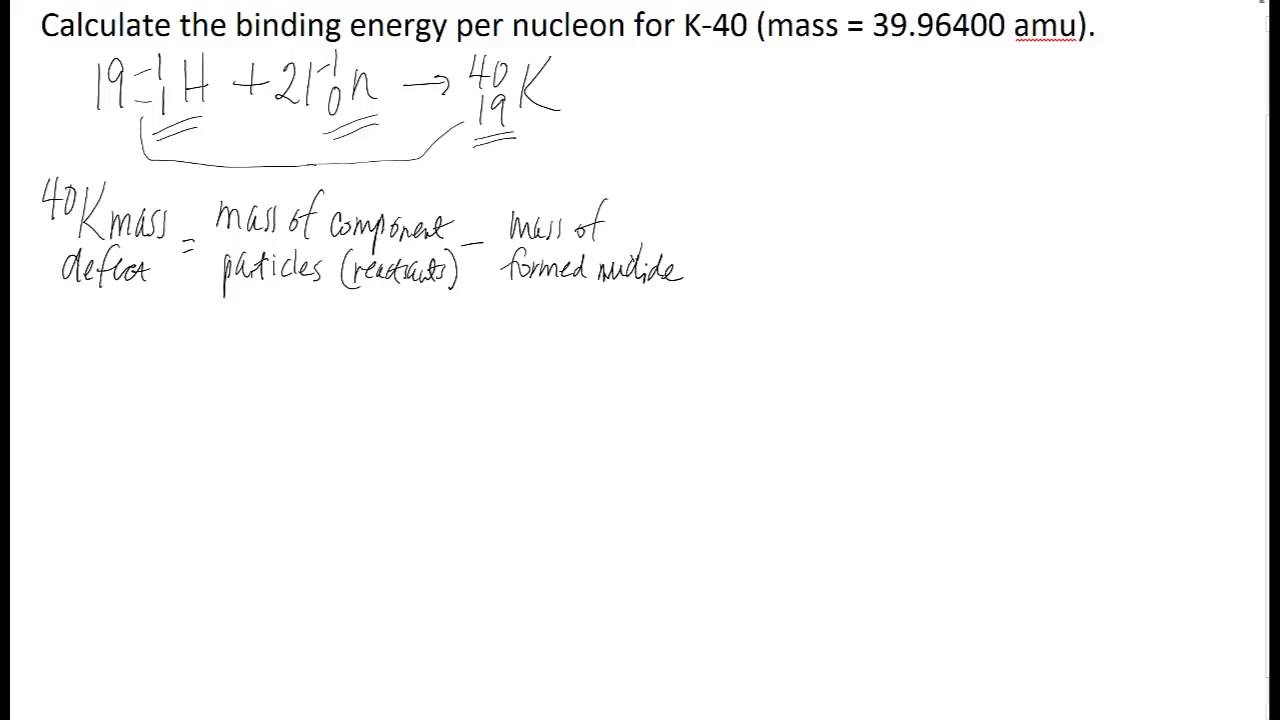
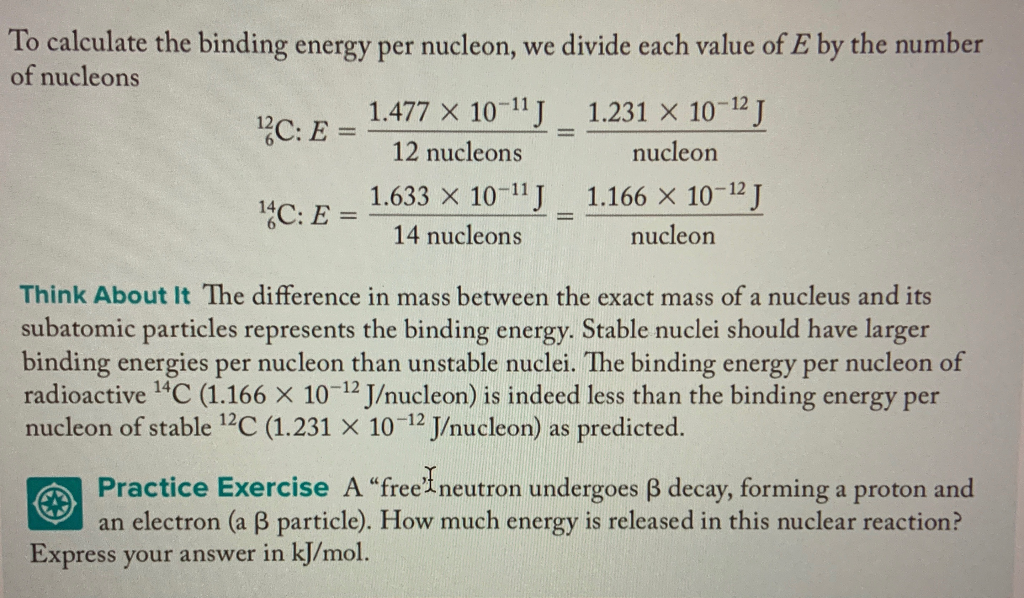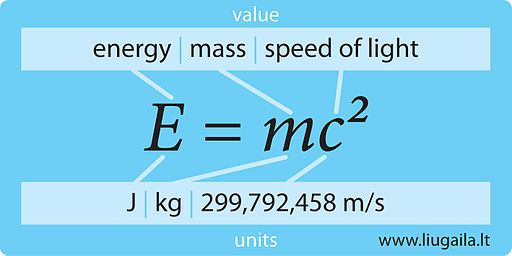Binding energy per nucleon in 2He4 is Given, Mass of 2He4 = 4.002604 amu Mass of proton = 1.007825 amu Mass of neutron = 1.008665 amu

homework and exercises - Calculating energy released by using binding energy VS mass difference - Physics Stack Exchange

How do you calculate the mass defect and nuclear binding energy per nucleon of each of the nuclides? | Socratic
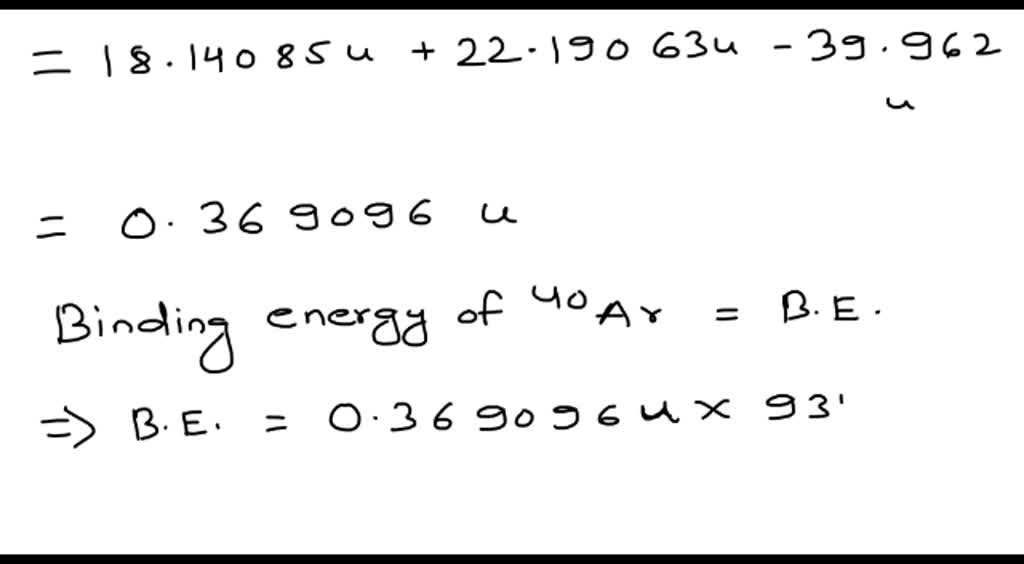
SOLVED:Calculate (in MeV) the total binding energy and the binding energy per nucleon (a) for ^40 Ar and (b) for ^40 K.