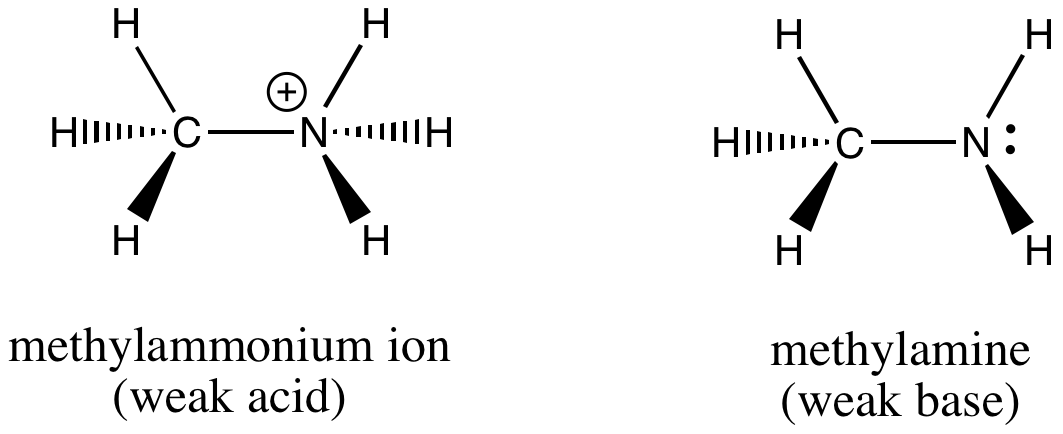
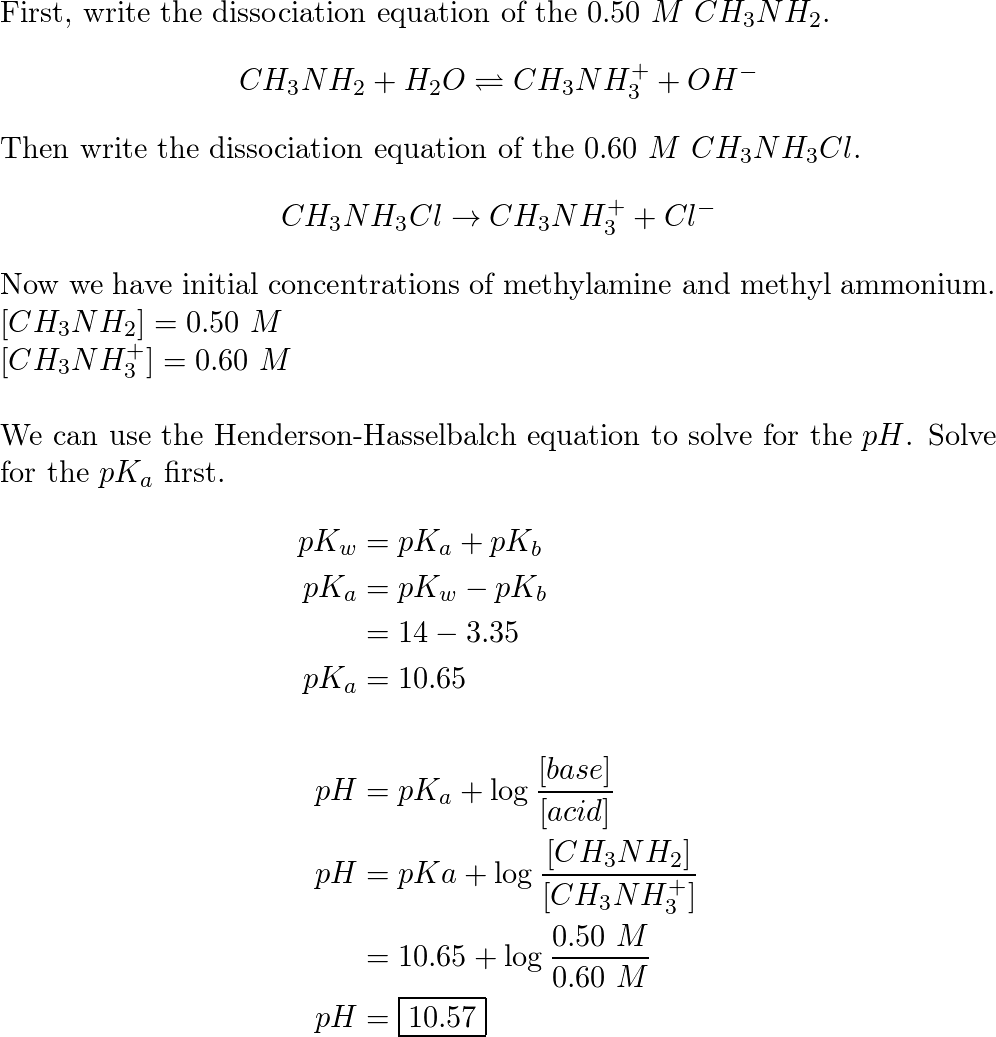How would you determine is the following salts will from a solution that is acidic, basic, or pH neutral? CH3NH3CN, Fe(ClO4)3, K2CO3, CH3NH3CL, RbI | Socratic

SOLVED: Consider 0.25 M solutions of the following salts. For each salt, indicate whether the solution is acidic, basic, or neutral. Ca(NO3)2 CH3NH3Cl NaCN C2H5NH3NO3 C3H7NH3I

Write the net ionic reaction that occurs upon the addition of hno3 to a solution which contains methylamine - Brainly.com

SOLVED: Which of the following compounds, when dissolved in water will produce an acidic solution? X. KNO3 Y. CH3NH3Cl Z. NaHSO4 X only X and Y Y and Z (Correct answer) Z

Solved) - Is the solution of KCI acidic, basic or neutral?. Is the solution... (1 Answer) | Transtutors










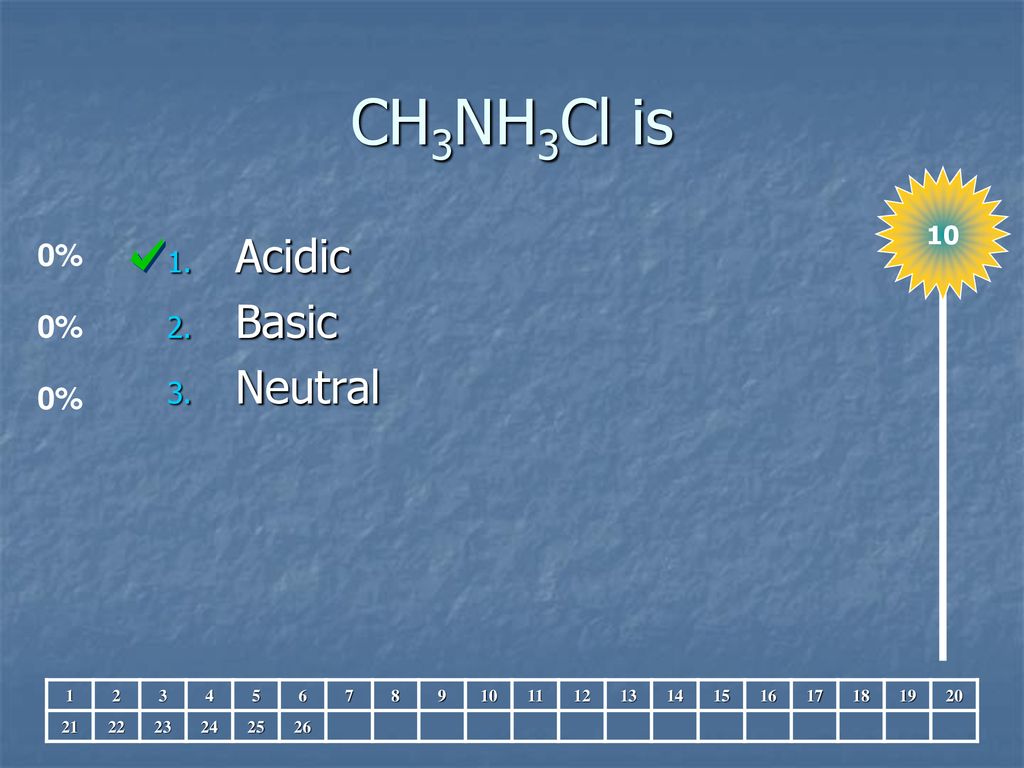
![14.82 | What is [OH−] in a solution of 0.125 M CH3NH2 and 0.130 M CH3NH3Cl? - YouTube 14.82 | What is [OH−] in a solution of 0.125 M CH3NH2 and 0.130 M CH3NH3Cl? - YouTube](https://i.ytimg.com/vi/rLbt_7_Q8r4/maxresdefault.jpg)