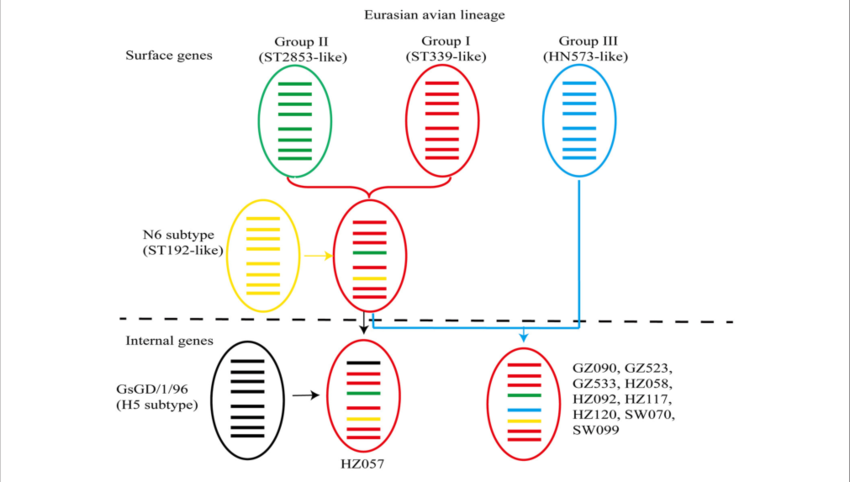
The origin of internal genes contributes to the replication and transmission fitness of H7N9 avian influenza virus | bioRxiv

Nonconcomitant host-to-host transmission of multipartite virus genome segments may lead to complete genome reconstitution | PNAS