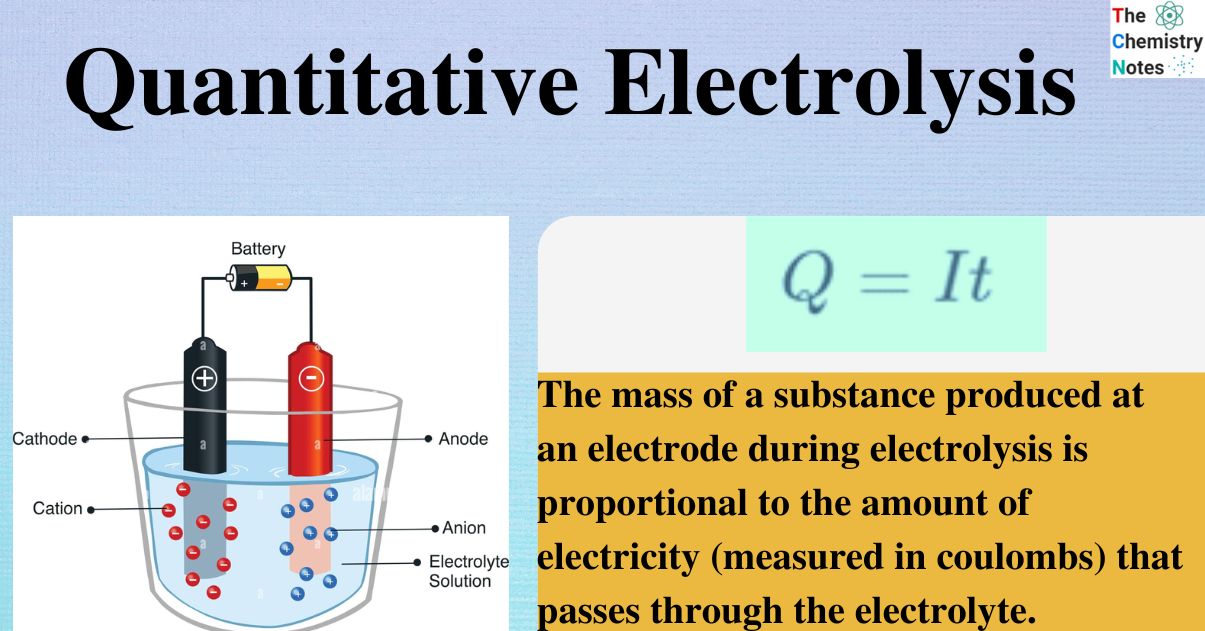
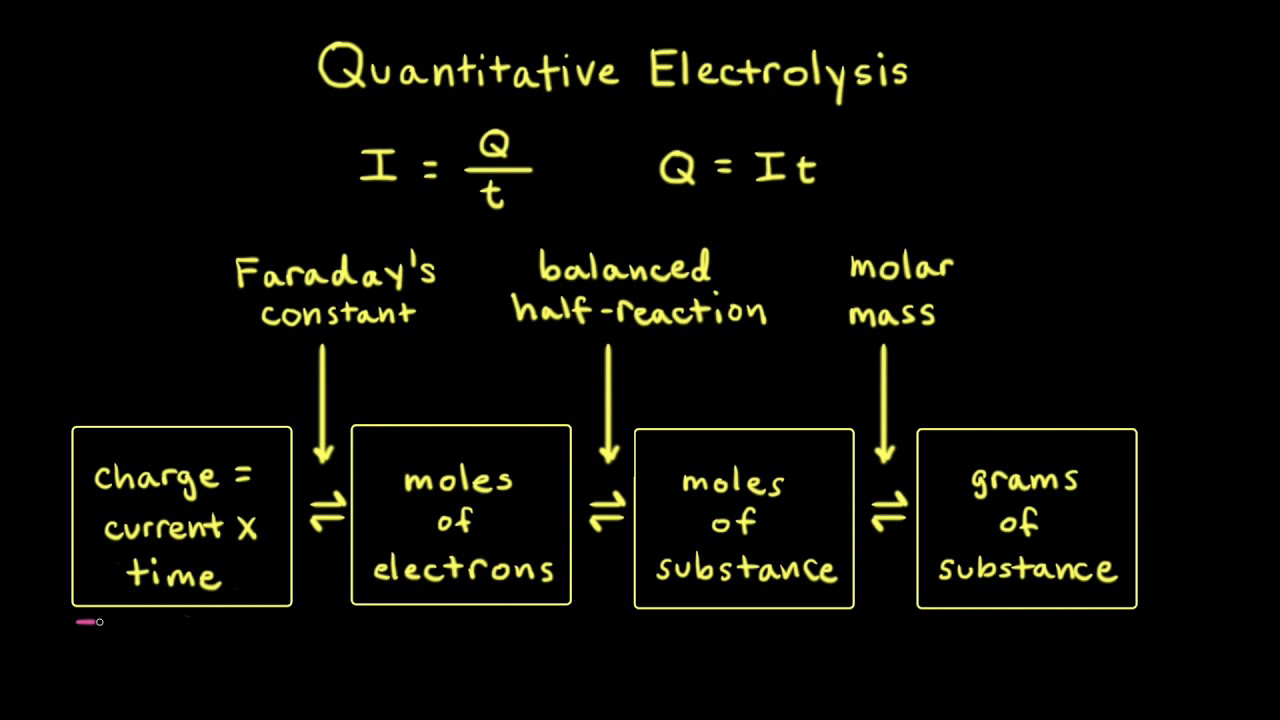
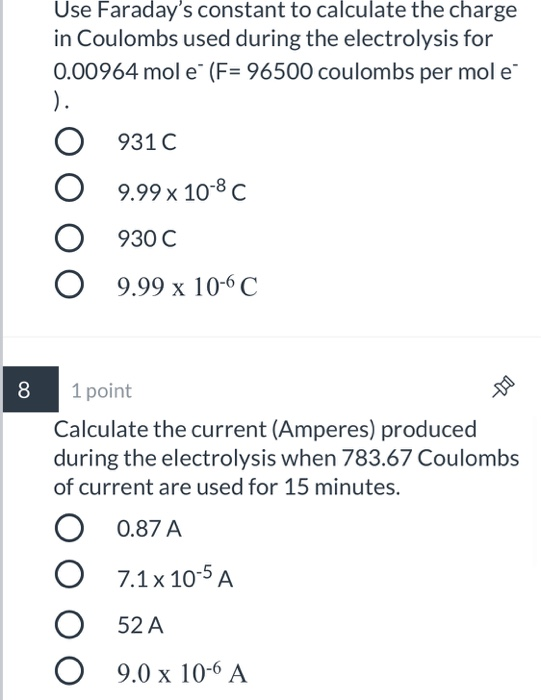
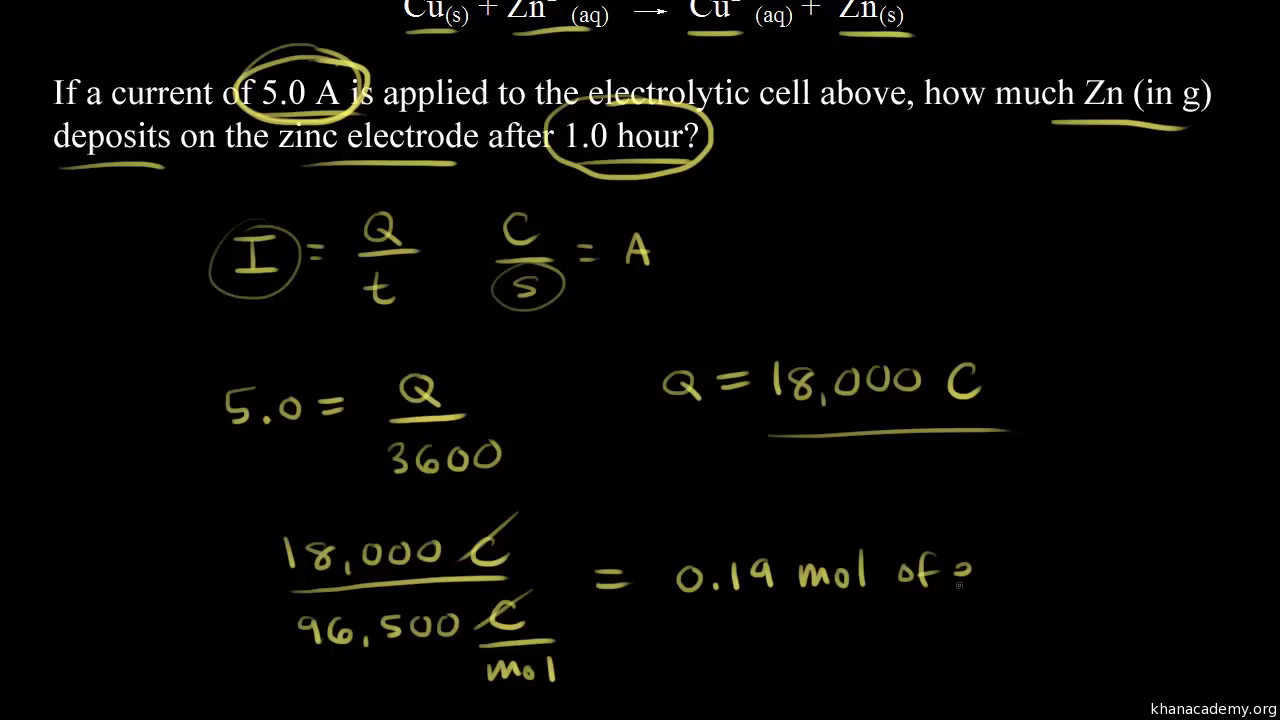Welcome to Chem Zipper.com......: During electro refining of Cu how much time is needed to produce 250g Cu on the cathode if the current is kept at 11 A?
Calculate volume of gases produced at stp at cathode and anode respectively during electrolysis of h2o by 10 faraday

Question Video: The Ionic Equation for the Electrolysis of the Molten Salt of Barium at the Negative Electrode | Nagwa

SOLVED: Electrolysis of aqueous solution of 0.1 M CuBrz was carried out using inert electrode at 25 Determine the products of electrolysis at anode and cathode: Justify. Calculate the mass of product
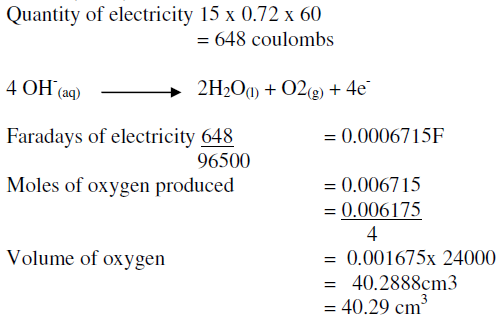
During the electrolysis, a current of 0.72A was passed through the electrolyte for 15 munites.Calculate the volume of gas produced at the anode.(1 Faraday = 96...