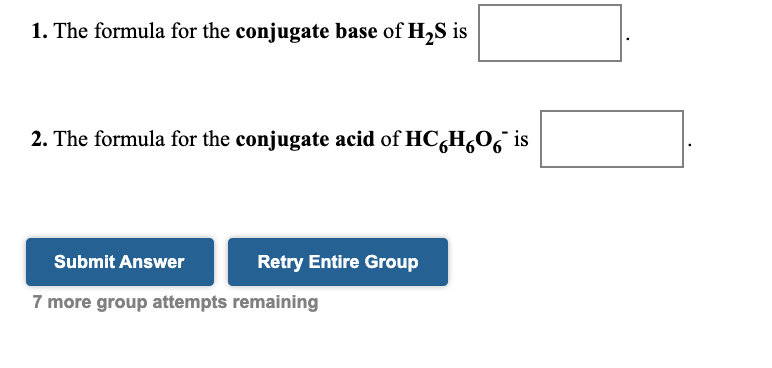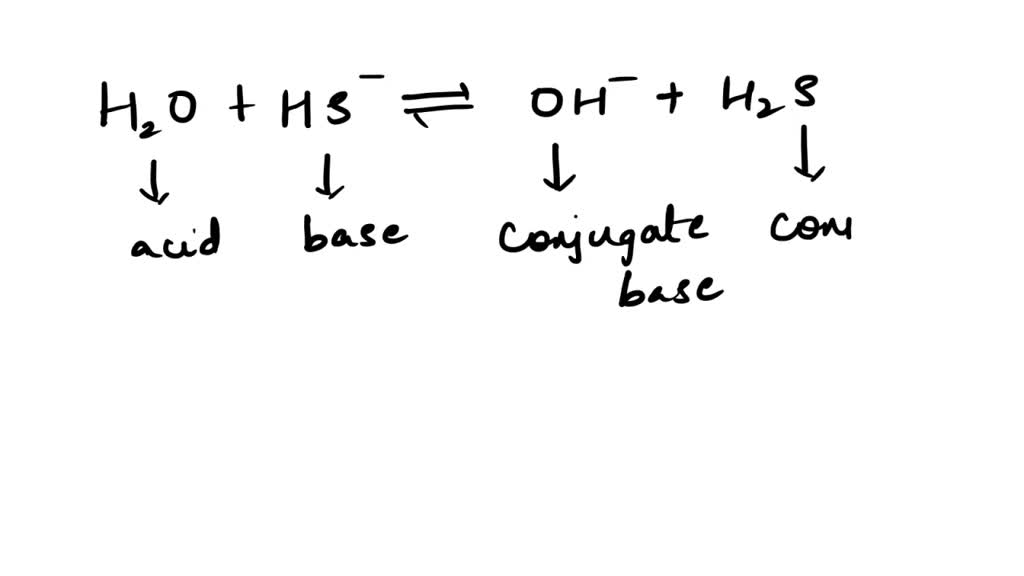
SOLVED: Be sure to answer all parts. In the following equation, identify the acids, bases, and conjugate pairs. H2O + HS− ⇌ OH− + H2S (a) What is the acid? H2O HS−

Hydrogen Sulfide: Chemical Biology Basics, Detection Methods, Therapeutic Applications, and Case Studies | Wiley

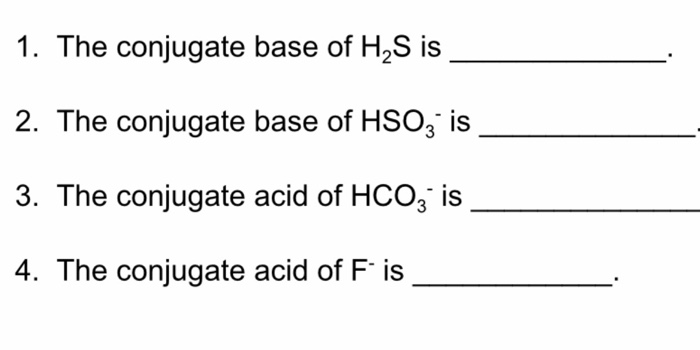
SOLVED: Identify the conjugate acid base pairs in each of the following: 1. HS-(aq) + H2O(l) ⇌ H2S(aq) + OH-(aq) 2. H2S(aq) + NH3(aq) ⇌ NH4(aq) + HS-(aq) 3. H2SO4(aq) + H2O(l)

SOLVED: Write an equation that shows the reaction of hydrogen sulfide, HS– with hydroxide ion, OH–. Label the acid, the base, the conjugate acid, and the conjugate base.
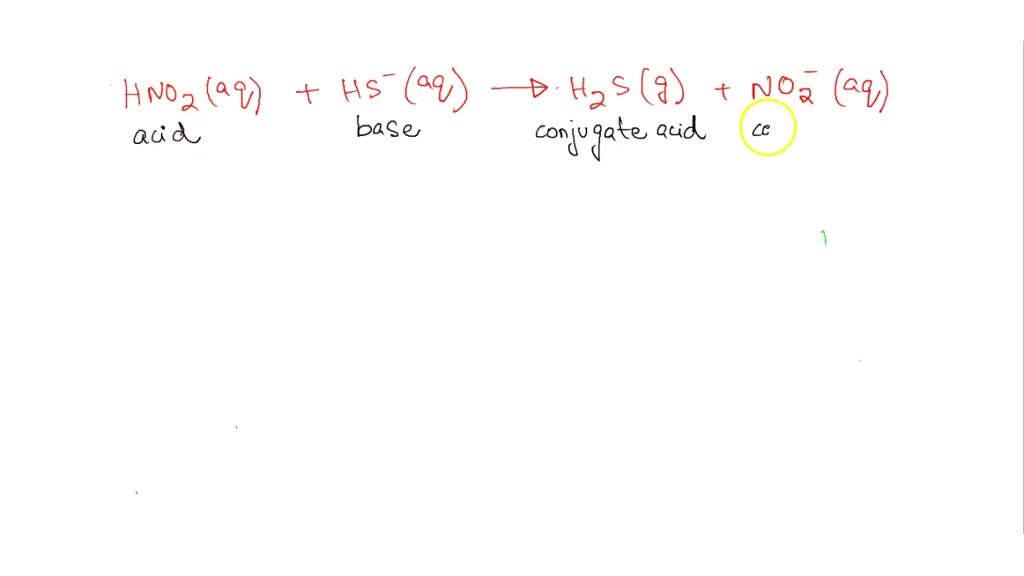
SOLVED: Consider the following reaction. Which are conjugate acid/base pairs? HNO2(aq) + HS-(aq) —-> H2S(g) +NO2-(aq) Group of answer choices H2S NO2^- HNO2 HS^- HS^- NO2^- HNO2 NO2^- HNO2 H2S
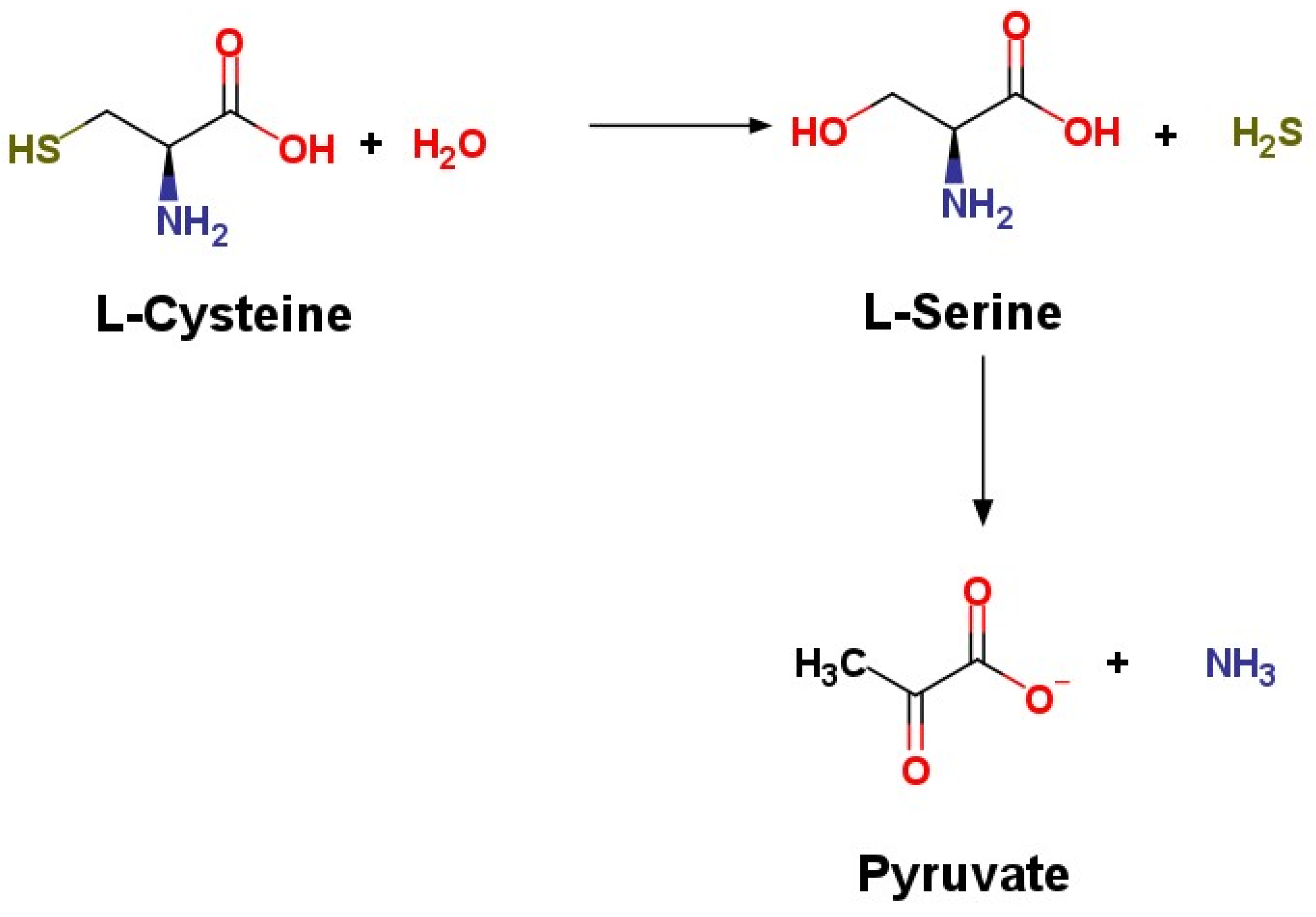
IJMS | Free Full-Text | Role of Hydrogen Sulfide, Substance P and Adhesion Molecules in Acute Pancreatitis
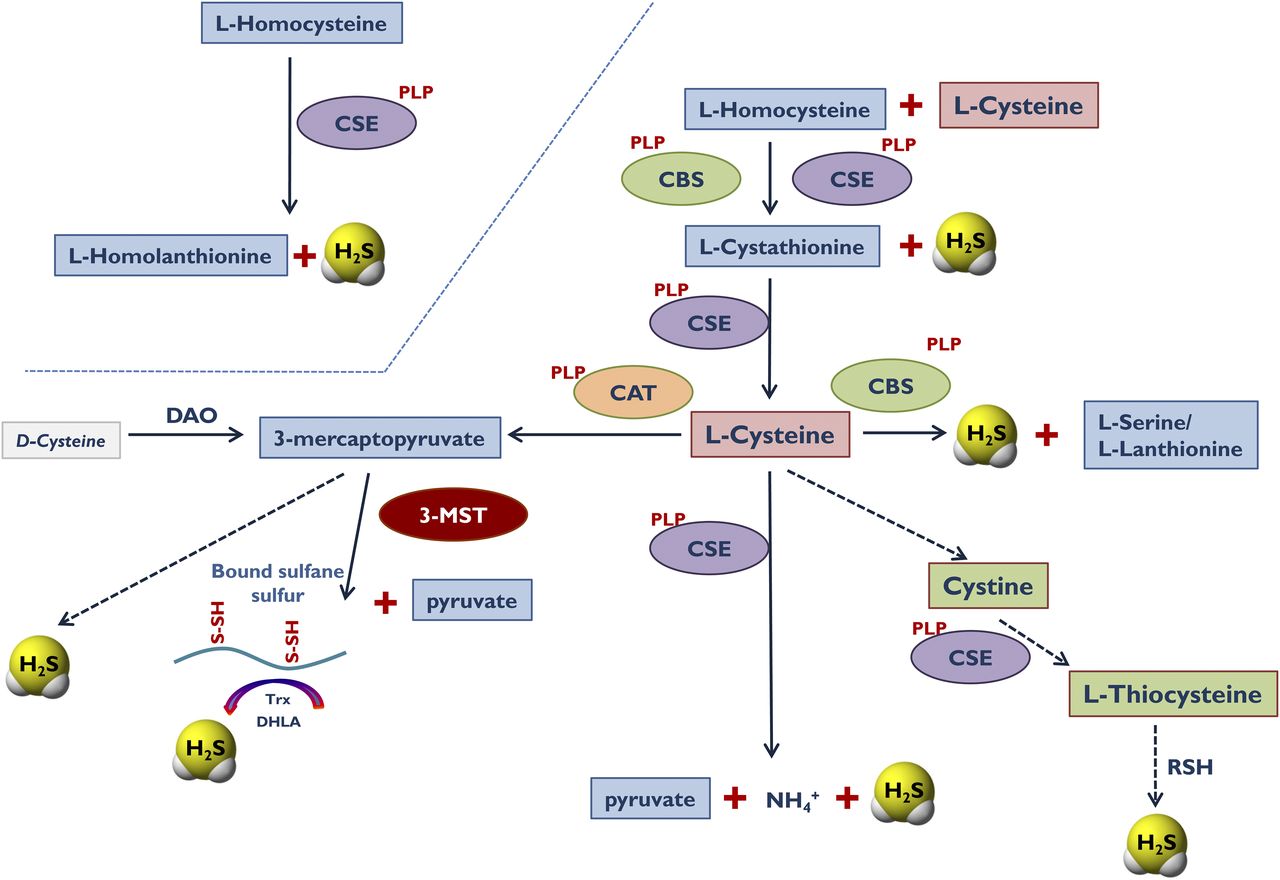











.jpg)