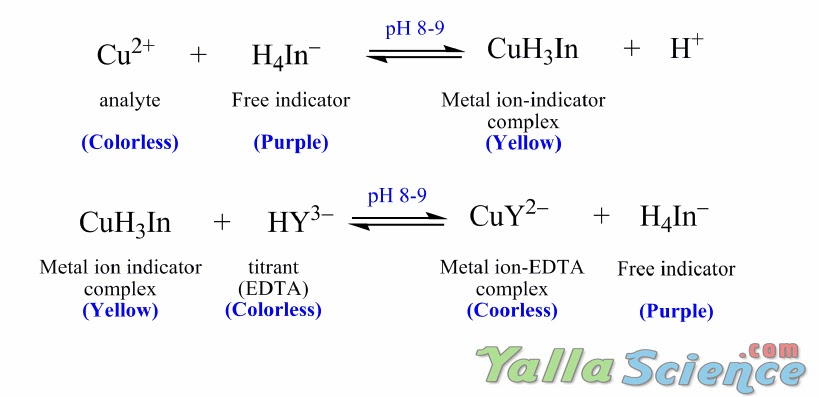
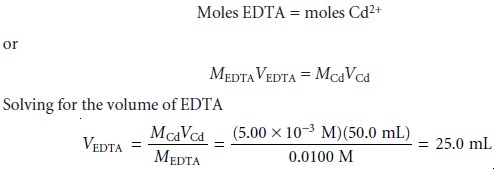SOLVED: DETERMINATION OF WATER HARDNESS BY EDTA TITRATION What is the basis of the *40000' in the equation? (mLof titrantsample)(M of titrant)(40000) Ca2+ (mg/,) mL of sample What should be the level

Understanding Complexometric Titrations of Metal Cations with Aminopolycarboxylic Acids (EDTA and Analogs) within the frame of the Notion of Reactions between Groups of Chemical Species

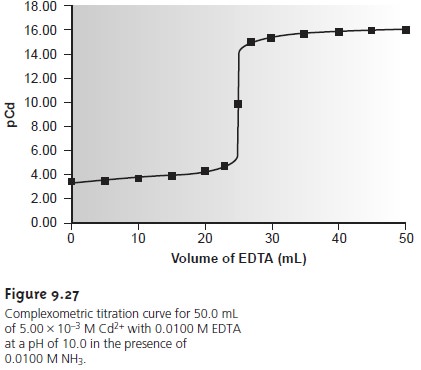
SOLVED: Complexometric Titration 10 ml of MgSO4 was titrated with 0.02M EDTA solution until end point appear (blue color) in the presence of 5.0 ml of ammonia buffer pH-I0 and 5 drops

SOLVED: 1) Use the molarity of the EDTA solution and the average volumeof EDTA added to calculate the average number of moles of EDTArequired for the titration. Molarity EDTA= .01 Volume EDTA= .